Rigidified Clicked Dimeric Ligands for Studying the Dynamics of the PDZ1‐2 Supramodule of PSD‐95
2014.11.18Eildal, J. N., Bach, A., Dogan, J., Ye, F., Zhang, M., Jemth, P., & Strømgaard, K. (2015). ChemBioChem, 16(1), 64-69.
PSD-95 is a scaffolding protein of the MAGUK protein family, and engages in several vital protein–protein interactions in the brain with its PDZ domains. It has been suggested that PSD-95 is composed of two supramodules, one of which is the PDZ1-2 tandem domain. Here we have developed rigidified high-affinity dimeric ligands that target the PDZ1-2 supramodule, and established the biophysical parameters of the dynamic PDZ1-2/ligand interactions. By employing ITC, protein NMR, and stopped-flow kinetics this study provides a detailed insight into the overall conformational energetics of the interaction between dimeric ligands and tandem PDZ domains. Our findings expand our understanding of the dynamics of PSD-95 with potential relevance to its biological role in interacting with multivalent receptor complexes and development of novel drugs.
- Recommend
-
2025-10-22
IQSEC2/BRAG1 may modulate postsynaptic density assembly through Ca2+-induced phase separation.
-
2025-08-22
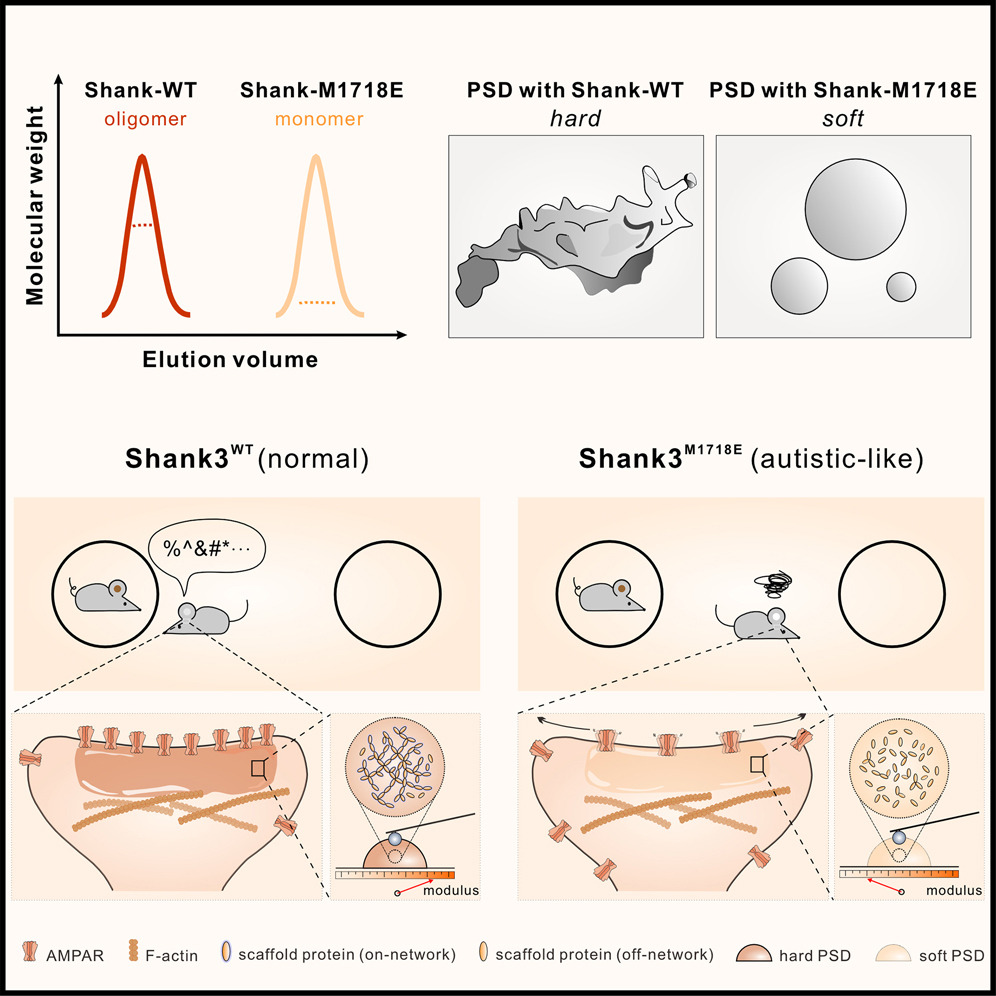
Shank3 oligomerization governs material properties of the postsynaptic density condensate and synaptic plasticity.
-
2025-08-21
Modulating synaptic glutamate receptors by targeting network nodes of the postsynaptic density condensate.
-
2025-08-19
Current practices in the study of biomolecular condensates: a community comment.
-
2025-06-10
Phase separation instead of binding strength determines target specificities of MAGUKs.