Synaptic targeting and function of SAPAPs mediated by phosphorylation-dependent binding to PSD-95 MAGUKs
2017.12.26Zhu, J., Zhou, Q., Shang, Y., Li, H., Peng, M., Ke, X., ... & Zhang, M. (2017). Cell reports, 21(13), 3781-3793.
The PSD-95/SAPAP/Shank complex functions as the major scaffold in orchestrating the formation and plasticity of the post-synaptic densities (PSDs). We previously demonstrated that the exquisitely specific SAPAP/Shank interaction is critical for Shank synaptic targeting and Shank-mediated synaptogenesis. Here, we show that the PSD-95/SAPAP interaction, SAPAP synaptic targeting, and SAPAP-mediated synaptogenesis require phosphorylation of the N-terminal repeat sequences of SAPAPs. The atomic structure of the PSD-95 guanylate kinase (GK) in complex with a phosphor-SAPAP repeat peptide, together with biochemical studies, reveals the molecular mechanism underlying the phosphorylation-dependent PSD-95/SAPAP interaction, and it also provides an explanation of a PSD-95 mutation found in patients with intellectual disabilities. Guided by the structural data, we developed potent non-phosphorylated GK inhibitory peptides capable of blocking the PSD-95/SAPAP interaction and interfering with PSD-95/SAPAP-mediated synaptic maturation and strength. These peptides are genetically encodable for investigating the functions of the PSD-95/SAPAP interaction in vivo.
- Recommend
-
2025-10-22
IQSEC2/BRAG1 may modulate postsynaptic density assembly through Ca2+-induced phase separation.
-
2025-08-22
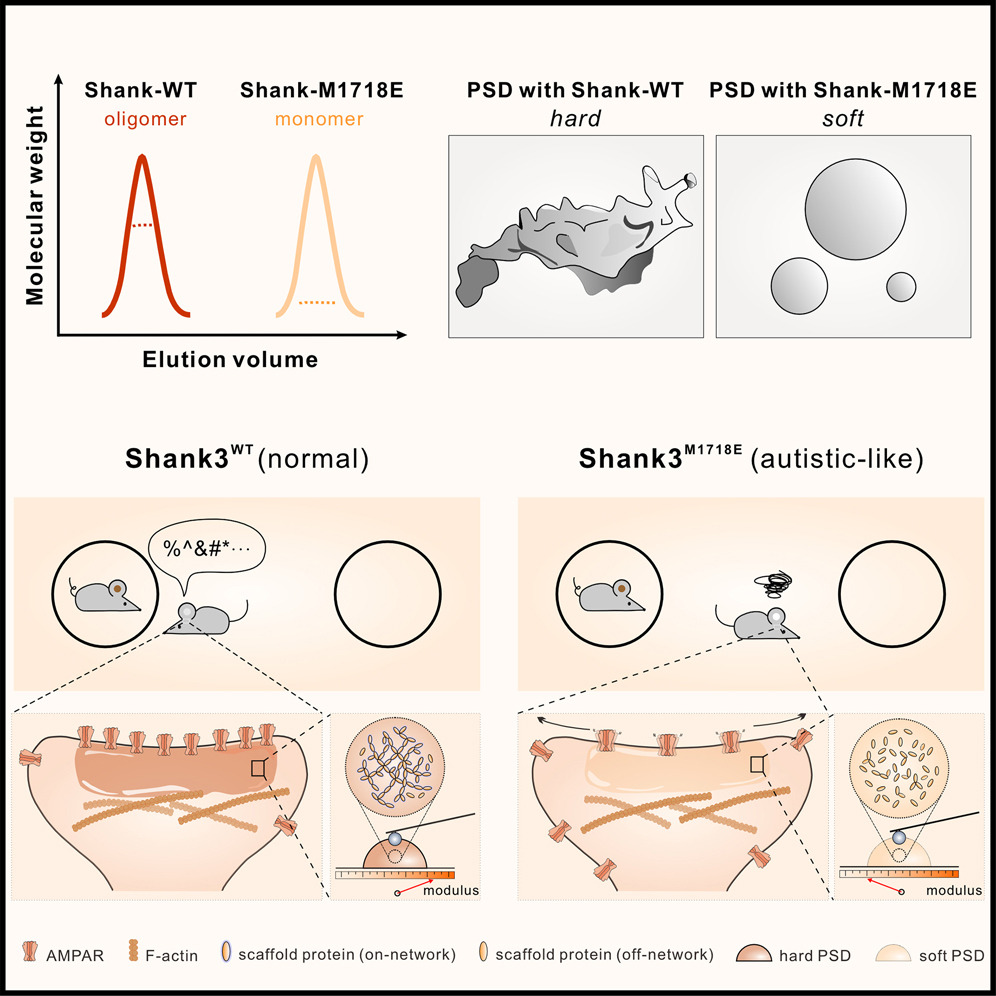
Shank3 oligomerization governs material properties of the postsynaptic density condensate and synaptic plasticity.
-
2025-08-21
Modulating synaptic glutamate receptors by targeting network nodes of the postsynaptic density condensate.
-
2025-08-19
Current practices in the study of biomolecular condensates: a community comment.
-
2025-06-10
Phase separation instead of binding strength determines target specificities of MAGUKs.