Phase separation instead of binding strength determines target specificities of MAGUKs.
2025.06.10Chen, Y., Ma, C., Shen, Z., Chen, S., Zhu, S., Jia, B., ... & Zhang, M. (2025). Nature Chemical Biology, 1-11.
Homologous proteins often have distinct functions, even if they share overlapping binding targets. PSD-95 and MAGI-2, two membrane-associated guanylate kinase (MAGUK)-family scaffolds in neuronal synapses, exemplify this. With unknown mechanisms, the two MAGUKs are localized at distinct subsynaptic compartments with PSD-95 inside the postsynaptic density (PSD) and MAGI-2 outside. Here we demonstrate that MAGI-2 forms condensates through phase separation. When coexisting with PSD proteins, the MAGI-2 condensate can enrich the extrasynaptic N-cadherin–β-catenin adhesion complex and the MAGI-2 condensates are immiscible with the PSD-95 condensates. Surprisingly, phosphorylated SAPAP is selectively enriched in the PSD-95 condensate, even though it binds to MAGI-2 with a higher affinity. The specific localization of SAPAP is because of the higher network complexities of the PSD-95-containing condensate than the MAGI-2 condensate. Thus, phase-separation-mediated molecular condensate formation can generate a previously unrecognized mode of molecular interaction and subcellular localization specificities that do not occur in dilute solutions.
- Recommend
-
2025-10-22
IQSEC2/BRAG1 may modulate postsynaptic density assembly through Ca2+-induced phase separation.
-
2025-08-22
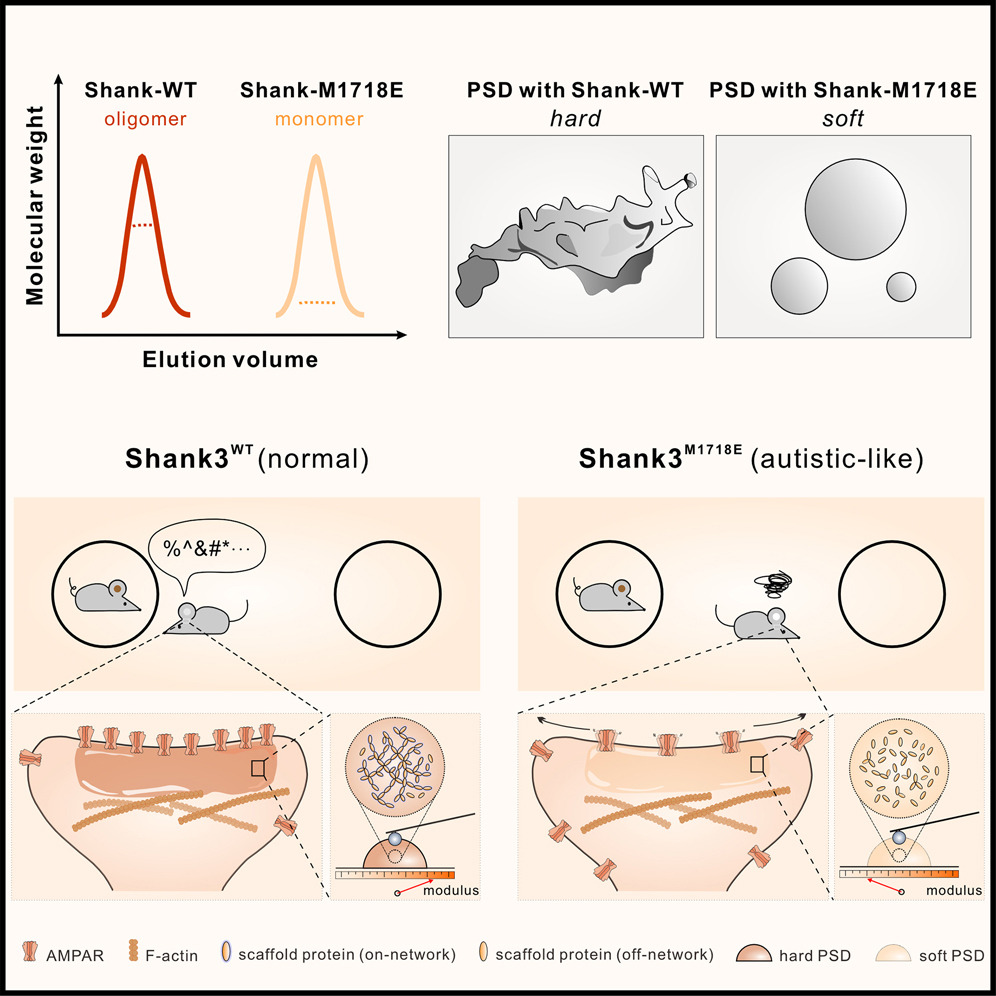
Shank3 oligomerization governs material properties of the postsynaptic density condensate and synaptic plasticity.
-
2025-08-21
Modulating synaptic glutamate receptors by targeting network nodes of the postsynaptic density condensate.
-
2025-08-19
Current practices in the study of biomolecular condensates: a community comment.
-
2025-06-10
Phase separation instead of binding strength determines target specificities of MAGUKs.