The structure of the ZMYND8/Drebrin complex suggests a cytoplasmic sequestering mechanism of ZMYND8 by Drebrin
2017.11.07Yao, N., Li, J., Liu, H., Wan, J., Liu, W., & Zhang, M. (2017). Structure, 25(11), 1657-1666.
Malfunctions of the actin binding protein Drebrin have been implicated in various human diseases such as Alzheimer's disease, cognitive impairments, cancer, and digestive disorders, though with poorly understood mechanisms. The ADF-H domain of Drebrin does not contain actin binding and depolymerizing activity. Instead, it binds to a histone marker reader, ZMYND8. Here we present the high-resolution crystal structure of Drebrin ADF-H in complex with the ZMYND8 PHD-BROMO-PWWP tandem, elucidating the mechanistic basis governing the highly specific interaction of the two proteins. The structure reveals that the ZMYND8 PHD-BROMO-PWWP tandem forms a structural supramodule that is necessary for binding to Drebrin ADF-H. Drebrin ADF-H competes with modified histone for binding to ZMYND8. Binding of Drebrin can shuttle ZMYND8 from nucleus to cytoplasm in living cells. Taken together, our study uncovers a non-actin target binding mode for ADF-H domains, and suggests that Drebrin may regulate activities of epigenetic reader ZMYND8 via its cytoplasmic sequestration.
- Recommend
-
2025-10-22
IQSEC2/BRAG1 may modulate postsynaptic density assembly through Ca2+-induced phase separation.
-
2025-08-22
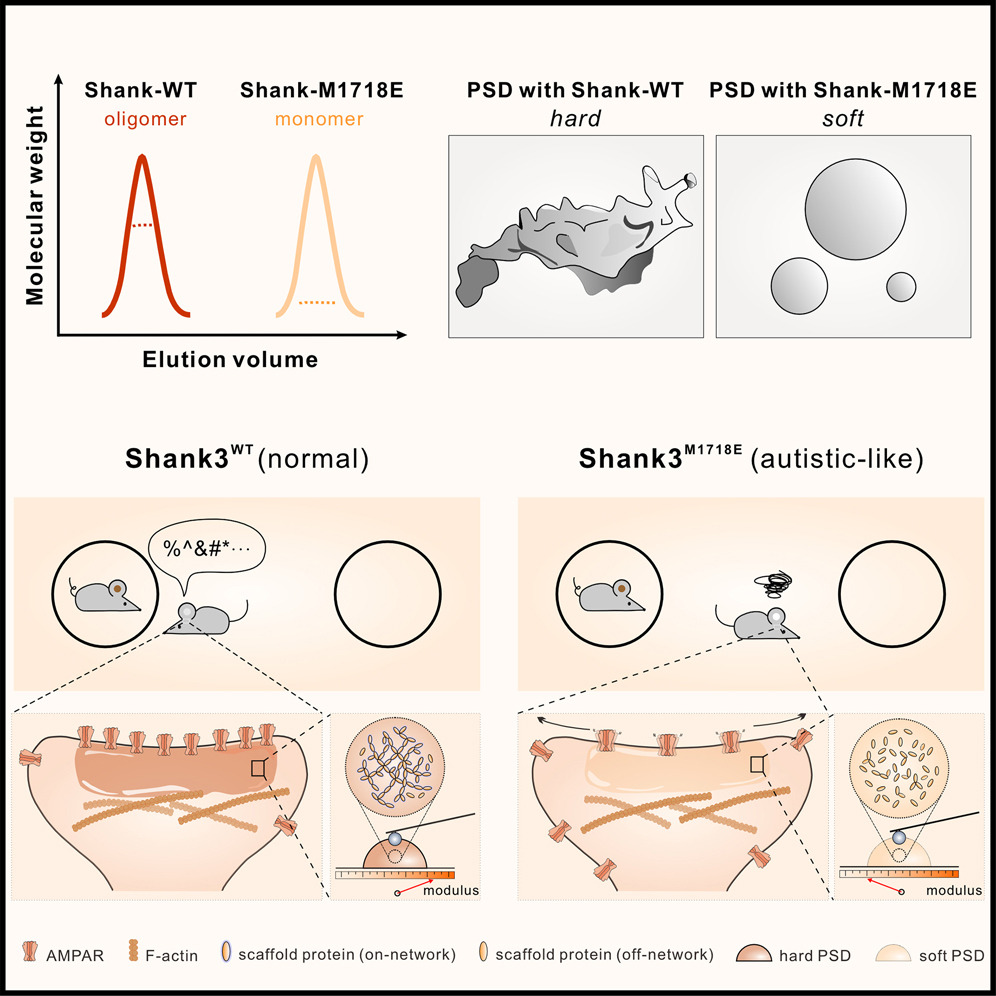
Shank3 oligomerization governs material properties of the postsynaptic density condensate and synaptic plasticity.
-
2025-08-21
Modulating synaptic glutamate receptors by targeting network nodes of the postsynaptic density condensate.
-
2025-08-19
Current practices in the study of biomolecular condensates: a community comment.
-
2025-06-10
Phase separation instead of binding strength determines target specificities of MAGUKs.