Assembly of model postsynaptic densities involves interactions auxiliary to stoichiometric binding
2022.01.04Lin, Y. H., Wu, H., Jia, B., Zhang, M., & Chan, H. S. (2022). Biophysical Journal, 121(1), 157-171.
The assembly of functional biomolecular condensates often involves liquid-liquid phase separation (LLPS) of proteins with multiple modular domains, which can be folded or conformationally disordered to various degrees. To understand the LLPS-driving domain-domain interactions, a fundamental question is how readily the interactions in the condensed phase can be inferred from interdomain interactions in dilute solutions. In particular, are the interactions leading to LLPS exclusively those underlying the formation of discrete interdomain complexes in homogeneous solutions? We address this question by developing a mean-field LLPS theory of two stoichiometrically constrained solute species. The theory is applied to the neuronal proteins SynGAP and PSD-95, whose complex coacervate serves as a rudimentary model for neuronal postsynaptic densities (PSDs). The predicted phase behaviors are compared with experiments. Previously, a three SynGAP/two PSD-95 ratio was determined for SynGAP/PSD-95 complexes in dilute solutions. However, when this 3:2 stoichiometry is uniformly imposed in our theory encompassing both dilute and condensed phases, the tie-line pattern of the predicted SynGAP/PSD-95 phase diagram differs drastically from that obtained experimentally. In contrast, theories embodying alternate scenarios postulating auxiliary SynGAP-PSD-95 as well as SynGAP-SynGAP and PSD-95-PSD-95 interactions, in addition to those responsible for stoichiometric SynGAP/PSD-95 complexes, produce tie-line patterns consistent with experiment. Hence, our combined theoretical-experimental analysis indicates that weaker interactions or higher-order complexes beyond the 3:2 stoichiometry, but not yet documented, are involved in the formation of SynGAP/PSD-95 condensates, imploring future efforts to ascertain the nature of these auxiliary interactions in PSD-like LLPS and underscoring a likely general synergy between stoichiometric, structurally specific binding and stochastic, multivalent “fuzzy” interactions in the assembly of functional biomolecular condensates.
- Recommend
-
2025-10-22
IQSEC2/BRAG1 may modulate postsynaptic density assembly through Ca2+-induced phase separation.
-
2025-08-22
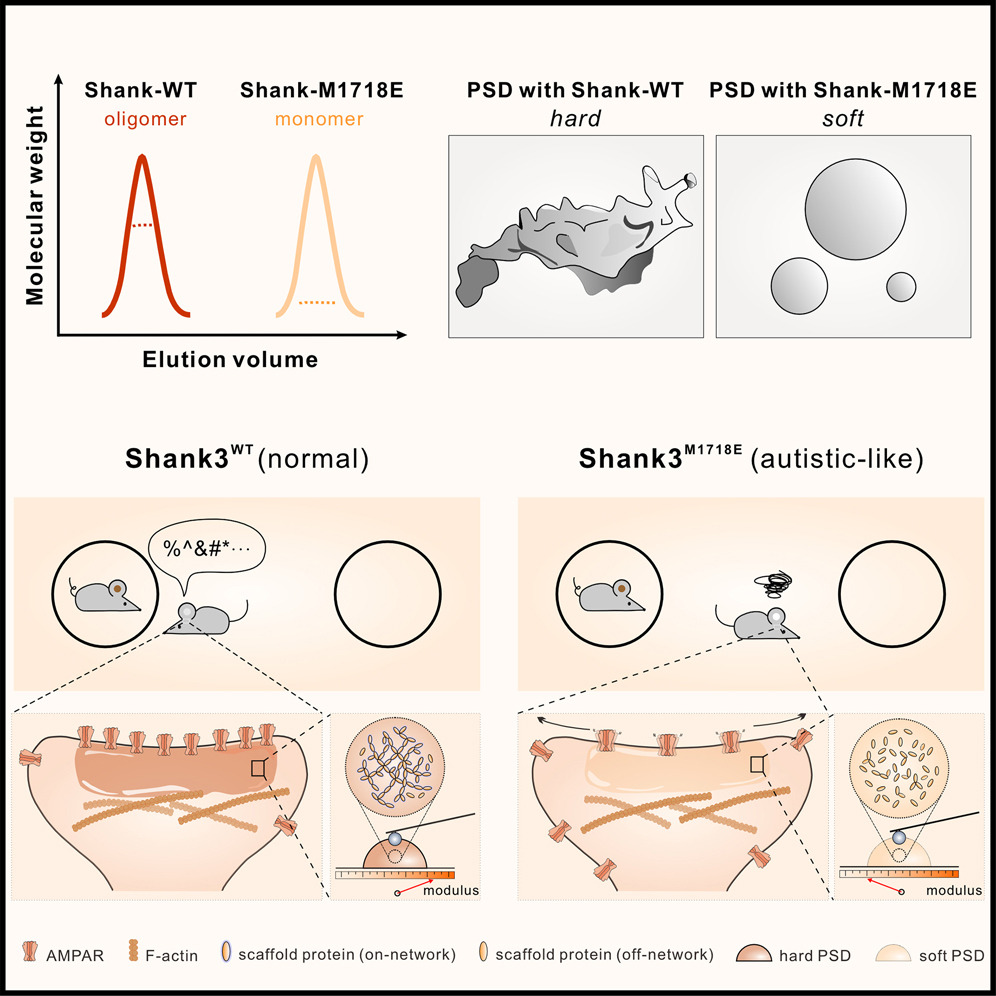
Shank3 oligomerization governs material properties of the postsynaptic density condensate and synaptic plasticity.
-
2025-08-21
Modulating synaptic glutamate receptors by targeting network nodes of the postsynaptic density condensate.
-
2025-08-19
Current practices in the study of biomolecular condensates: a community comment.
-
2025-06-10
Phase separation instead of binding strength determines target specificities of MAGUKs.